Biacore Molecular Interaction Shared Resource
Contact Information
Aykut Üren, MD., Director
Telephone: (202) 687-9504
au26@georgetown.edu
E312 Research Building
Purushottam Tiwari, PhD, Associate Professor
pbt7@georgetown.edu
Telephone: (202) 687-3841
E320 Research Building

Overview
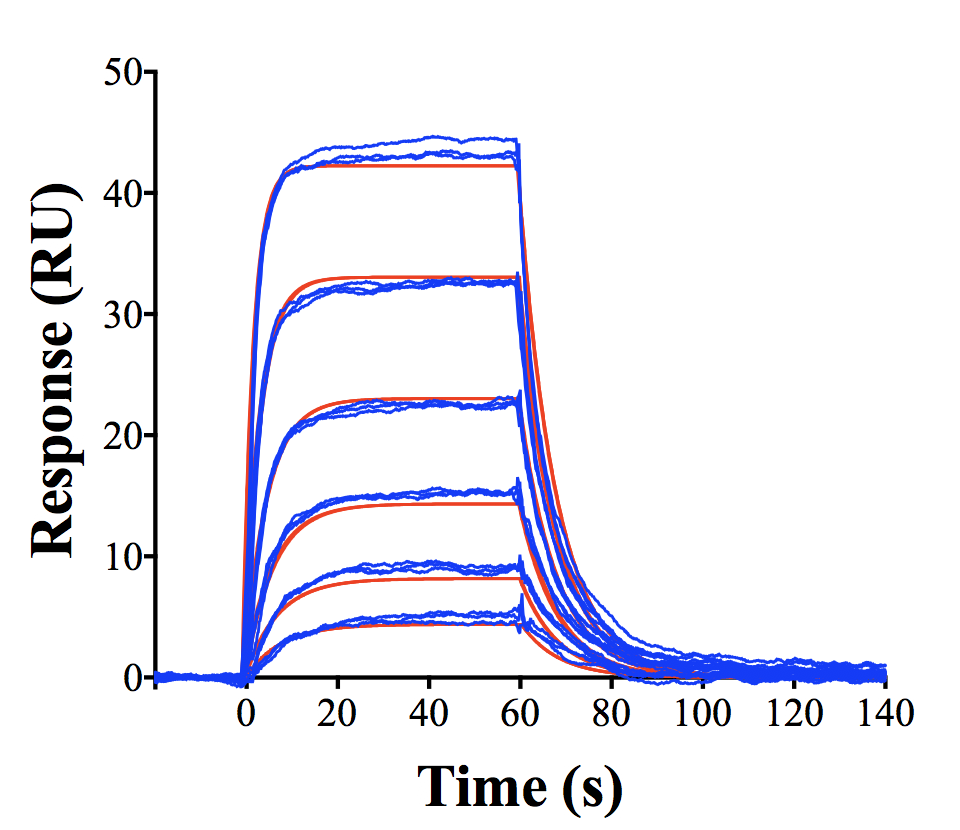
The Biacore Molecular Interaction Shared Resource (BMISR) provides customized biomolecular analysis services to Georgetown University faculty and staff. The two Biacore machines in our facility, Biacore T-200 and Biacore 4000, utilize the Surface Plasmon Resonance (SPR) technology to study molecular binding events on a chip surface. The basic principle involves immobilization of a ligand on a sensor chip followed by delivery of an analyte by a microfluidic system. Any protein, DNA, RNA, lipid, carbohydrate, polysaccharide, cell, virus, drug, drug like molecule (organic or inorganic) can be used as the ligand or analyte. Since the detection system is based on measuring total mass on the sensor chip surface neither ligand nor analyte has to be tagged.
In addition to identifying binding partners to a target molecule, SPR also provides quantitative data on:
- Specificity: How specific is the binding between two molecules?
- Concentration: How much of a given molecule is present and active?
- Kinetics: What is the rate of association and dissociation?
- Affinity: How strong is the binding?
Location
E312 Research Building, Georgetown University
Shipping address:
Attn: Biacore, Research Building Room E312
Georgetown University Medical Center
3970 Reservoir Rd NW
Washington, D.C. 20057-1469
